Overcoming Solubility Challenges: Techniques to Improve Dissolution Rate and Increase Bioavailability
Developing formulations where the active pharmaceutical ingredient (API) is sufficiently bioavailable is a central focus for pharmaceutical scientists, so improving dissolution of poorly soluble actives is a key consideration during the drug product development process.
In this article, our Director of Drug Delivery Technologies, Kimberly Zubris, discusses the formulation of insoluble compounds, outlining techniques to enhance bioavailability and incorporate these APIs into successful drug products.
Pre-Formulation Groundwork
Pre-formulation groundwork gives us a greater understanding of the properties of an API, which aids in development and provides a more streamlined pathway to safe and effective formulations.
The stability characteristics of a compound impact how it will be processed. For example, it is important to know the extent to which an API can withstand stressors, such as heat and light. When a compound is not thermally stable, processing techniques that require higher temperatures (such as hot melt extrusion) should be avoided. If an API is light sensitive, then we must set up the appropriate environmental conditions to protect it from UV exposure during processing and packaging. At Particle Sciences, we employ amber lighting in both our formulation and manufacturing spaces when dealing with light-sensitive compounds.
Solubility Challenges
There are a growing number of APIs in development that exhibit poor water solubility, leading to low bioavailability and limited therapeutic effect. These molecules cannot be simply converted into a final dosage form such as an intravenous solution or an oral pill or tablet—they require the use of advanced formulation techniques to progress through the development pipeline.
A common model for describing solubility and permeability of APIs is the Biopharmaceutical Classification System (BCS), which divides molecules into four quadrants based on their behavior in a pre-defined aqueous environment (Figure 1). APIs that fall under a BCS Class II designation suffer from low solubility but exhibit high tissue permeability—meaning they are ideal candidates for solubility enhancement. BCS Class IV molecules—which exhibit low solubility and low permeability—require a combination of solubility enhancement and permeation enhancement to achieve a therapeutic effect.
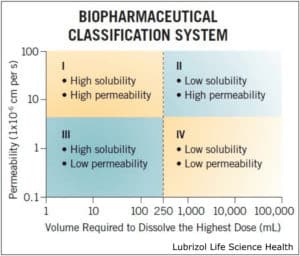
Figure 1: The biopharmaceutical classification system, which classifies APIs based on solubility and permeability.
Although finding a method to formulate new BCS Class II and IV molecules is challenging, there are existing techniques to improve solubility, increase bioavailability, and convert these APIs into viable drug products.
Formulation Techniques
Below are some common techniques for overcoming limited drug solubility and producing a finished product that has high bioavailability and therapeutic effect:
- Amorphous solids – Amorphous solid dispersions utilize techniques such as hot melt extrusion or spray drying to incorporate an API into a polymer matrix and maintain it in an amorphous form. As an amorphous material, the API may exhibit improved aqueous solubility and therefore higher bioavailability.
- Cyclodextrin complexes – Cyclodextrins are predominantly used as complexing agents to help enhance the aqueous solubility of poorly soluble actives. The chemical structure of a cyclodextrin molecule resembles a ring consisting of a hydrophobic “pocket” and a hydrophilic outer surface. The inner pocket is able to form an inclusion complex with lipophilic molecules, which shields the hydrophobic functionality of an API, while the hydrophilic outer surface of the cyclodextrin ensures the overall complex is water-soluble. Complexation with cyclodextrin often leads to improved aqueous solution stability and enhanced bioavailability for several routes of administration.
- Nanosuspensions – Nanosuspensions consist of nano-sized drug particles dispersed in an aqueous vehicle. These formulations are generated through nanomilling—a top-down approach to reducing the particle size of an API that has been used to formulate numerous commercial drug products. The basic principle behind nanomilling is increasing the surface area-to-volume ratio of an API by reducing the particle size below 1000 nm, typically in the 100s of nm range. Smaller particles exhibit greater interaction with water, which increases dissolution rate and improves bioavailability. Nanosuspensions have been converted into a wide range of dosage forms, including oral liquids, capsules, tablets, films, injectables, aerosols, and more.
Nanomilling: A Data-Driven Approach
At Particle Sciences, we view nanomilling as a nearly universal solubility enhancement approach, because it can be applied to just about any insoluble molecule, and formulation work can begin with only a small amount of API. In addition to its universality, nanomilling is also a robust, scalable process that can produce large quantities of consistent material when properly optimized. However, while the nanomilling process may be reproducible, developing a reliable nanosuspension formulation is not always a straightforward task. Without stabilization, nanomilled APIs tend to agglomerate or increase in particle size over time through a process called Ostwald Ripening. To overcome these challenges, stabilizers must be added to formulations. Stabilizer selection is an iterative process that relies on a combination of experience, pre-formulation data, and a complete understanding of stabilizer properties to be successful. Particle Sciences is an industry leader in nanomilling for solubility enhancement, with decades of experience developing formulations for a range of APIs and dosage forms.
Other approaches to enhance solubility include encapsulation techniques, pH modification, and generation of salt forms. At Particle Sciences, we utilize all these solubility enhancement methods, relying on our years of formulation expertise to help our clients find the right solution for their API.
The Future
The rise in discovery of insoluble APIs over the past decade has put increased emphasis on techniques to increase bioavailability. With these molecules presenting formulation challenges, it’s essential to explore more diverse formulation techniques so that we can provide patients with effective drug products.
While there are various approaches that can be used to increase bioavailability, expertise, and experience are required to develop viable and effective formulations. Employing an intelligent approach to drug development helps to overcome the challenges of limited solubility and advance APIs through the development pipeline. To continue driving the pharmaceutical industry forward, Particle Sciences remains data-driven and open to various formulation techniques during drug product development. This approach, coupled with reliable analytical services and production support, will ensure the future success of our clients and the pharmaceutical industry as a whole.
Authors: